
Mercury Analysis Systems
In our previous blog post “What is Cold Vapor Atomic Absorption (CVAA) Spectroscopy?” (HERE), we discussed CVAAS as a primary technique for mercury analysis and determination. In this post, we will describe Teledyne Leeman Labs’ mercury analyzers (analysers outside of the US and Canada) that use the CVAAS process.
Leeman Labs offers two CVAA systems for mercury determination: the Hydra IIAA , which is an entry-level CVAA system that can detect mercury limits of less than 5.0 ng/L with a usable range of 5 ng/L to 1 mg/L; and the QuickTrace® M-7600 CVAA mercury analyzer, which is a dual-beam system capable of ultra-trace detection limits of less than 0.5 ng/L.
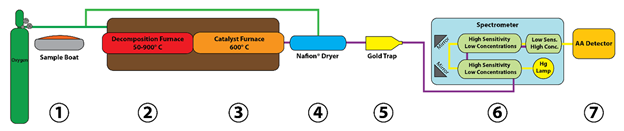
Hydra IIAA Mercury Analyzer
The Hydra IIAA mercury analyzer design is based on US EPA method 245.1, which allows for determination of total mercury in drinking, surface, ground, sea and brackish waters and industrial and domestic wastewaters. Method 245.1 typically requires sample digestion techniques that produce liquid samples with mercury present in the +2 valence state. The working process of the Hydra IIAA analyzer is detailed below.
First an acidified, digested, aqueous sample from an autosampler is introduced via the peristaltic pump as Hg2 dissolved in a solution. At the same time, a reducing agent (10% SnCl2 in 7 - 10% HCl) is introduced via another parallel peristaltic pump channel. A sodium or potassium borohydride solution may also be used in place of stannous chloride (SnCl2).
The sample and the reagent streams meet at the mixing tee and then enter the mixing tube where the stannous chloride (Sn2+) reduces Hg2+ into Hg0, while the mixture is enroute to the Gas Liquid Separator (GLS). At this stage, the analyte is present as a finely dispersed emulsion of liquid (metallic) Hg0 micro-droplets, in excess SnCl2 solution medium. Next, the carrier gas sweeps the mercury vapor through the Nafion™ Dryer and to the detector.
Visit our website for more information on the Hydra IIAA Mercury Analyzer.
It is also notable that the Hydra IIAA system can be converted to a direct analysis combustion mercury analyzer by quickly changing the analyzer’s module. Conversion to a combustion mercury analyzer allows customers the flexibility to measure mercury in solid samples without pretreatment or digestion.
For more information on the Hydra IIAA mercury analyzer, refer to the product overview page on our website (HERE). For a comparison of Teledyne’s Leeman Labs’ mercury analyzer features, download the “Selecting the Best Technique for Mercury Measurement - A Practical Guide” using the button below. This helpful guide covers our entire catalog of mercury analyzers and provides insight into which mercury analyzer is best for your laboratory. Our instrumentation covers multiple atomic spectroscopy techniques including atomic fluorescence spectroscopy.
QuickTrace M-7600 CVAA Mercury Analysis System
The QuickTrace® M-7600 mercury analyzer design is based on the US EPA method 245.1, but provides the ability to also analyze samples using performance-based US EPA 245.7. US EPA 245.7 was developed to address state-based needs for measuring toxic metals at ambient water quality criteria (WQC) levels, to protect designated uses. Both methods typically require sample digestion techniques that produce liquid samples with mercury present in the +2 valence state.
The carrier gas (Ar or N2) enters the reference cell of the spectrometer to facilitate measurement of the incident radiant power (P0) at 253.7 nm, and then continues on to the GLS. Meanwhile, an acidified digested aqueous sample from the autosampler is introduced via the peristaltic pump as Hg2+ dissolved in solution. Simultaneously, a reducing agent (10% SnCL2 in 7% HCl) is introduced via a parallel peristaltic pump channel. The sample and reagent (SnCl2) streams join at the mixing tee, and immediately enter the liquid mixing tube. Stannous chloride (Sn2+) reduces Hg2+ in solution to Hg0 while the mixture is enroute to the GLS. At this stage the analyte is present as a finely dispersed emulsion of liquid (metallic) Hg0 micro-droplets, in excess SnCl2 solution. This vapor is swept by the carrier gas through the Nafion™ dryer and into the sample cell of the dual-beam spectrometer.
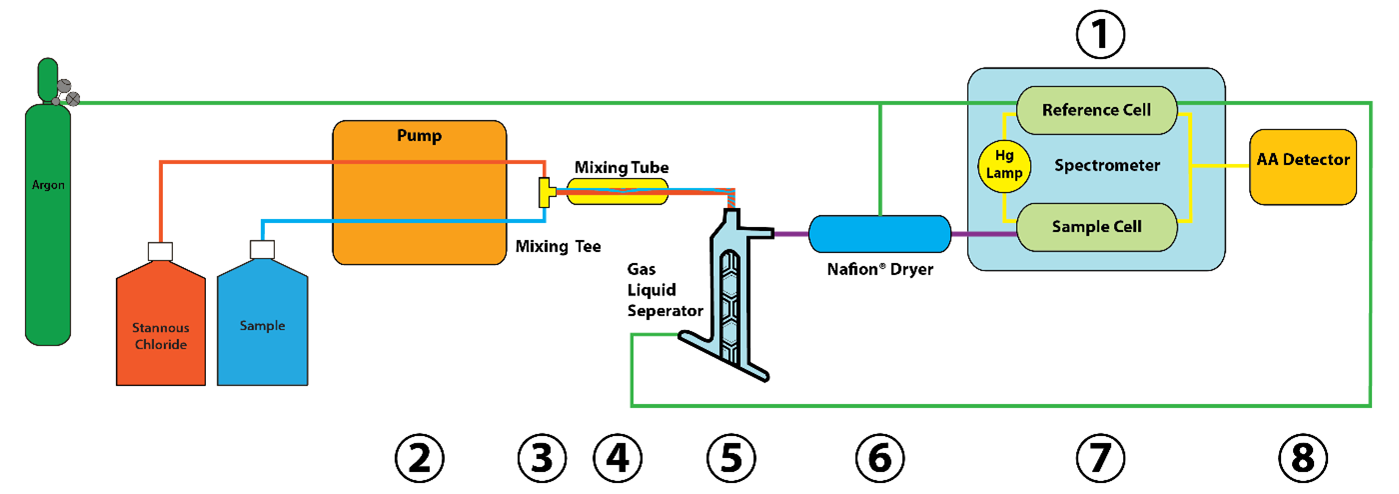
For more information on the QuickTrace® M-7600 mercury analyzer, refer to the product overview page on our website (HERE). For a comparison of Teledyne’s Leeman Labs’ mercury analyzer features, download the “Selecting the Best Technique for Mercury Measurement - A Practical Guide” using the button below. This helpful guide covers our entire catalog of mercury analyzers and provides insight into which mercury analyzer is best for your laboratory. Our instrumentation covers multiple atomic spectroscopy techniques including atomic fluorescence spectroscopy.
Visit our website for more information on the QuickTrace® M-7600 CVAA Mercury Analyzer
For more details about how the QuickTrace M-7600 works beyond this stage, review Teledyne’s Practical Guide for Selecting the Best Technique for Mercury Measurement.
FAQs
What does a mercury analyzer measure?
Mercury analyzers are capable of detecting mercury (Hg) and quantifying its concentration, in solids, liquids and gases. Knowledge of mercury’s presence and the amount that is present is useful for protecting the health and safety of plants, animals, humans and the environment. The Hg in the sample is often determined by atomic spectroscopy. The two types of atomic spectroscopy are atomic absorption (AA) and atomic fluorescence (AF). These methods measure the quantity of light that is absorbed or emitted at a wavelength specific for mercury, 253.7nm. The amount of light absorbed or released can be used to calculate the amount of the element in the sample.

